In this article we will discuss about:- 1. Types of Immune Responses 2. Phases of Immune Responses 3. Pathways.
Types of Immune Responses:
The collective and coordinated responses of different components of the immune system to the introduction of foreign substances are called the immune response.
In general, the vertebrate immune system produces two major types of immune responses:
(1) Humoral immune response and
ADVERTISEMENTS:
(2) Cell-mediated immune response (CMI). Both types of immune responses are elicited by various lymphocytes, the main component of adaptive immune system. Thus, these responses are all adaptive immune responses giving adaptive immunity to the host.
1. Humoral Immune Response (HIR):
The adaptive immune response mediated by antibodies produced by B-lymphocytes (plasma cells) is referred to as humoral immune response. Since antibodies are found in the plasma, lymph and tissue fluid, i.e., in the body fluids (also called humors), this immunity as well as immune response are referred to as humoral immunity and humoral immune response, respectively.
It protects the organism against extracellular microbes and toxins secreted by them and foreign macromolecules.
2. Cell-mediated Immune Response:
This form of adaptive immune response is mediated by the T-lymphocytes and gives immunity against microbes that survive within phagocytes or infected non-phagocytic cells.
ADVERTISEMENTS:
Intracellular microbes like viruses and some bacteria, survive and proliferate within phagocytes and other host cells, where they are inaccessible to circulating antibodies. Cell- mediated immunity can recognise and eliminate such intracellular microbes.
Phases of Immune Responses:
All immune responses are initiated by the recognition of foreign antigen by specific lymphocytes. This recognition, in turn, may lead to activation of the lymphocyte that recognises the antigen as well as culminate in the development of effector mechanisms.
It then mediates the physiological function of the immune response, i.e., the elimination of the antigen. Once the antigen is eliminated, the immune response abates and homoeostasis (previous condition) is restored.
Thus the overall immune response may be divided into following phases:
1. Recognition of Antigens:
ADVERTISEMENTS:
It is really surprising to understand that how the immune system of an individual respond to a large number and variety of antigens. According to clonal selection hypothesis, each individual possesses antigen-specific clone of lymphocytes which develop before the exposure to any antigen.
The cells constituting each clone have identical antigen receptors, which are different from the receptors present on the cells of all other clones. These receptors are expressed at a stage and site of maturation where the lymphocytes have not encountered any antigens that is naive lymphocytes.
It is difficult to count or assess the number of antigenic determinant that can be recognised by the mammalian immune system.
However, it is estimated to be 107 to 109, the approximate number of different antigen receptor proteins that are produced. Therefore, it reflects the number of distinct clones of lymphocytes present in each individual. Foreign antigens interact with the pre-existing clones of lymphocytes in the specialised lymphoid organs where immune responses are initiated.
2. Activation of Lymphocytes:
The naive lymphocytes are activated by two distinct signals, the first is the antigen and second is either microbial products or components of innate immune responses to microbes. This is called two signal hypothesis.
However, the second signal is different in humoral immunity and cell-mediated immunity. The activation of lymphocyte consists of synthesis of new proteins, cellular proliferation and differentiation into effective and memory cells.
3. Effector Phase:
During this phase, antigens are eliminated. Antibodies and T- lymphocytes with the help of other effector cells and defence mechanism eliminate extracellular and intracellular microbes.
4. Homeostasis:
After elimination of antigens, the immune system returns to resting state, i.e., the immune responses are declined. At this time most progenies of the antigen-stimulated lymphocytes die by apoptosis. The survival of the lymphocytes is dependent on antigen and antigen-induced growth factors. As the immune response eliminates the antigen that initiated it, the lymphocytes become deprived of essential survival stimuli.
Pathways of Immune Responses:
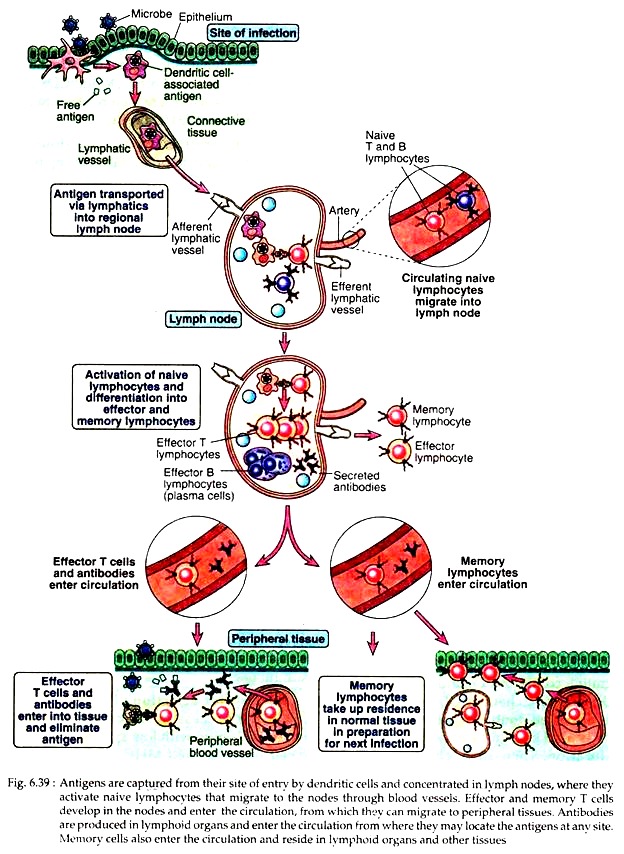
Key events that are generated during immune responses may be illustrated as follows (see Fig. 6.39):
1. T Cell and Cell-Mediated Immunity:
The phases of adaptive immune response (in general) have been discussed previously. Different populations of T cells differ in their requirements for activation and in the pattern of their responses. Here the general features of T cell maturation, activation and cell-mediated immune responses will be focused.
A. Maturation of T cell:
The maturation of T lymphocytes from committed progenitors follows sequential stages consisting of:
(i) Somatic recombination and expression of TCR genes,
(ii) Cell proliferation,
(iii) Thymic selection, and acquisition of mature phenotypes and functional capabilities.
(i) Somatic recombination and expression of TCR genes:
T lymphocytes originate from precursors that arise in the foetal liver and adult bone marrow. In the eighth or ninth week of gestation in humans, the progenitor T cells migrate to thymus from the early sites of haematopoiesis.
Developing T cells in thymus are called thymocytes. The most immature cortical thymocytes contain TCR genes in their germ line configuration and do not express TCR, CD3 or Ϛ chain or CD4 or CD8; these cells are called double-negative thymocytes.
This is also known as pro-T cell stage of maturation. Then, through a complex rearrangement of TCR genes, these double negative cells form a novel complex called the pre-T cell receptor. Thus far, no pre-T cell analogous to the pre-B cell has been discovered, although, its existence has been hypothesized.
However, this receptor can recognise some intra-thymic ligand and transmit the signal. Such signal transduction induces expression of both CD4 and CD8 on the thymocytes which are then referred to as double positive (CD4+8+) cells.
(ii) Cell proliferation:
The double positive cells begin to proliferate. During this proliferation, TCR α chain gene rearrangement does not occur even in the presence of active RAG-1 and RAG-2 genes (RAG-1 and RAG-2 proteins are produced by RAG-1 and RAG-2 genes, respectively and are required for TCR gene rearrangement). The α gene cannot rearrange because the RAG-2 protein is rapidly degraded in proliferating cells.
So α- chain gene rearrangement does not begin until the double positive thymocytes stop proliferating and RAG-2 protein level increases. As a result, a clone of cells is generated with a single TCR β-chain rearrangement. Each cell of this clone can then rearrange different α-chain genes allowing for greater T cell diversity.
Double positive cells expressing both CD3 and the αβ T cell receptor begin to appear at day 17 and reach maximal levels about the time of birth. Only a small number of these cells, again through TCR gene rearrangement, develop either into single positive CD4+ or single positive CD8+ cells. These single positive cells migrate to the periphery of the thymus.
(iii) Thymic selection:
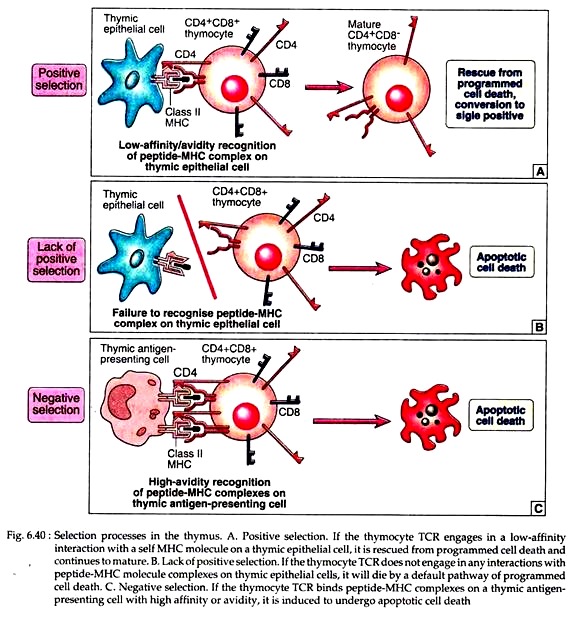
In the thymus, the developing T cells undergo two selection processes that are responsible for preserving useful cells and eliminating potentially harmful ones:
(a) Positive selection:
Positive selection is the process in which thymocytes whose TCRs bind with low avidity (weakly) to self-peptide – self MHC complexes are stimulated to survive (Fig. 6.40A). Thymocytes whose receptors do not recognise self MHC molecules are permitted to die by a default pathway of apoptosis (Fig. 6.40B).
This selection ensures that the T cells that mature are self MHC-restricted. It also fixes the class I or class II MHC restriction of T cell subsets, ensuring that CD8+ T cells are specific for peptides displayed by class 1 MHC molecules and CD4+ T cells for class II associated peptides.
(b) Negative:
Negative selection is the process in which thymocytes whose TCRs bind strongly to self-peptide antigens in association with self MHC molecules are deleted (Fig. 6.40C). This selection eliminates developing T cells that are strongly auto-reactive against ubiquitous self-antigens, most likely to be present in the thymus.
The net result of this selection process is that the repertoire of mature T cells that leave the thymus is self MHC restricted and tolerant to many self-antigens. A large and persuasive body of evidence demonstrated that some peptides play an important and central role in both positive and negative selections.
According to the transformation of double positive precursor into one of two different single positive lineages, two models have been proposed. The instructional model proposes that the multiple interactions between the TCR, CD8+ or CD4+ co-receptors and class I or class II MHC molecules instruct the cells to differentiate into either CD8+ or CD4+ single positive cells, respectively.
The second model, called the stochastic model, suggests that CD4 or CD8 expression is switched off randomly with no relation to the specificity of the TCR. Only those thymocytes whose TCR and remaining co-receptors recognise the same class of MHC molecule will mature.
B. Activation of T cell:
Professional antigen presenting cells, e.g., dendritic cells first capture the protein antigens that have entered through the epithelia and into the circulation. The captured antigens are then transported to lymph nodes and spleen (Fig. 6.39). Naive T cells continuously recirculate through the lymph nodes. When a T cell with its receptor finds the specific antigen, in the form of peptide-MHC-complex, that T cell is activated.
Interaction of a T cell with antigen initiates a cascade of biochemical events that induce the resting T cells to enter cell cycle (G0 to G1). When a TH cell with its receptor interact with a specific antigen bound with MHC II molecule complex, many of its genes are activated and as a result following products could be produced.
i. Immediate genes are expressed within half of an hour of antigen encounter that encode a number of transcription factors, viz, c-Fos, c-Myc, c-Jun, NF-AT etc.
ii. Early genes are expressed within 1-2 hour and encode IL-2, IL-2R, IL-3, IL-6, IFN-y etc.
iii. Late genes are expressed more than 2 days after antigen’s recognition and encode various adhesion molecules.
Signals for Activation:
All the above-mentioned molecules generate intracellular signals necessary for T cell activation. However, it is now known that this interaction, by itself is not sufficient to fully activate naive T cells. In fact, naive T cells require two distinct signals for activation and subsequent proliferation into effector cells:
(i) The initial signal (signal 1) is generated by interaction of an antigenic peptide with TCR-CD3 complex.
(ii) A subsequent co-stimulatory signal (signal 2) is provided by interaction between CD28 on the T cell and members of B7 family on the APCs. The principal co-stimulatory molecules expressed on APCs are the glycoproteins B7-1 and B7-2. Dendritic cells possess both these molecules.
Clonal selection and clonal anergy:
Presence or absence of the co-stimulatory factors determines whether a TH activation will result in clonal expansion or clonal anergy. If a resting TH cell receives the TCR-mediated signals (signal 1) in absence of a suitable co-stimulatory signal, then the TH cell will become anergic, i.e., it will not proliferate.
If both signal 1 and signal 2 are present, then resting TH cell will undergo G1 state of cell cycle resulting clonal expansion or cell proliferation.
C. Differentiation of T cell:
Most of the peripheral T cells express the αβ T cell receptors-CD3 complex. In the periphery there are about twice as many CD4+ T cells as CD8+ T cells. In general CD4+ cells function as T helper cells and CD8+ cells as cytotoxic cells. Both CD4+ and CD8+ T cells leave the thymus and enter the circulation as resting cells in G0 stage of the cell cycle.
These naive T cells continually recirculate between the bloods, lymph systems and may reside in secondary lymph nodes. If naive T cells do not encounter antigens in a lymph node, they exit through efferent lymphatics to thoracic duct and rejoin the blood.
A large scale recirculation of naive T cells actually increases the chance of encountering antigens. Naive T cells may survive about 5-7 weeks or more in the absence of antigen-stimulated activation.
After 48 hours of activation, the naive T- cells enlarge into a blast cell and begin undergoing repeated rounds of cell division producing various effector and memory cells. The T cells secrete its own growth promoting cytokines and also express cell surface receptors for these cytokines. This is called autocrine growth response. The principal autocrine growth factor is IL-2.
The cells that recognise antigen, bind the IL-2 they secrete and then proliferate in response to this cytokine. Another cytokine called IL-15 is produced by APCs, stimulates the proliferation of CD8+ T cells.
Before exposure to antigens, the number of naive T cells specific for any antigen is 1 in 105 to 106 lymphocytes. But after exposure to antigen, the number of T cells specific for that antigen may increase to about 1 in 10 for CD8+ cell and 1 in 100 to 1000 for CD4+ T cells.
The various effector cells carry out specialised functions. The CD4+ effector T cells produce two subsets (TH1 subset and TH2 subset) that produce distinct cytokines and perform different functions. The TH1 subset secretes IL-2, IFN-y and TNF-β and is responsible for classic cell-mediated functions, such as delayed-type hypersensitivity and the activation of cytotoxic T cells.
The TH2 subset secretes IL-4, IL-5, IL-6 and IL-10 and functions more effectively as a helper for B cell activation. The CD8+ cells differentiate into functional cytotoxic T-lymphocytes (CTLs).
Memory T cells are generally thought to be long-lived, antigen-activated T cells that respond with heightened reactivity to a subsequent challenge with the same antigens, generating a secondary response. Recent evidences suggest that some memory T cells may be short-lived cells maintained in the population by continued activation by persisting antigens.
In general, memory T cells express the same membrane molecules as effector cells. But, unlike the naive T cells that are activated only by dendritic cells, memory T cells can be activated by macrophages, dendritic cells and B cells.
D. Decline of T cell response:
T cell responses decline after the antigen is eliminated by effector cells. In fact, the elevated population of effector cells no longer benefits the host once the immune response has eliminated the pathogen.
Apoptosis acts to remove the expanded T cell population and eliminate activated T cells. The death of the TH cells is very useful because the high levels of cytokines produced by them are no longer needed and are even potentially deleterious to the host.
2. B cell and Humoral immunity:
The development of B cell involves several complicated steps. B lymphocytes arise continually throughout the life by differentiation from bone-marrow stem cells. There are many intermediate stages. The earliest recognisable B cells having cell surface receptor proteins are called pro B cell.
Then through sequential Ig-gene rearrangement these cells transform into pre B cell with membrane bound antibody consisting of µ heavy chain. During next stages of development, the pre B cells are transformed into virgin or naive B cells expressing complete IgM and IgD antibodies on membrane.
A. Activation of B cell:
The naive B cells are expelled from the bone marrow into the periphery where they may live only a few days as circulating cells, unless antigen interacts with their surface antibody, triggering further growth and maturation.
Virgin B cells may also enter the peripheral lymphoid organs (see pathway). Protein antigens are recognised by specific B and T cells in specific areas within the peripheral lymphoid organs. The activated cell populations then come together in these organs to initiate humoral responses.
Within 1-2 days after antigen administration, naive CD4+ T cells recognise the antigens presented by APCs like dendritic cells in the T cell zones of lymphoid organ. The T cells then leave the zones. At the same time, B cells recognise the antigen in the follicles and are activated and start to migrate toward the T cell zone.
Thus, an interaction between B and T cells occur at the interface of the follicles and T cell zones of lymphoid organs.
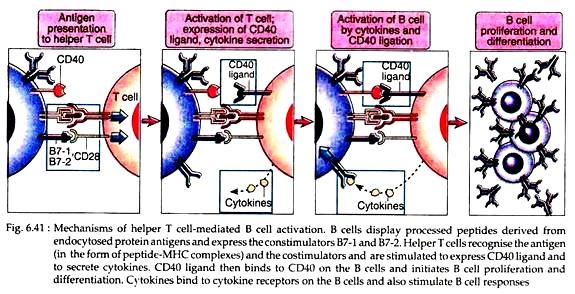
The B cells, in the meantime, degrade the captured protein antigen to peptides and display them on its surface in association with MHC class II molecules, just like the APCs. The achievement of B cell activation now requires the help of TH cells. The TH cells previously stimulated by MHC II antigen complex then recognise the peptides on the B cell surface and become further stimulated.
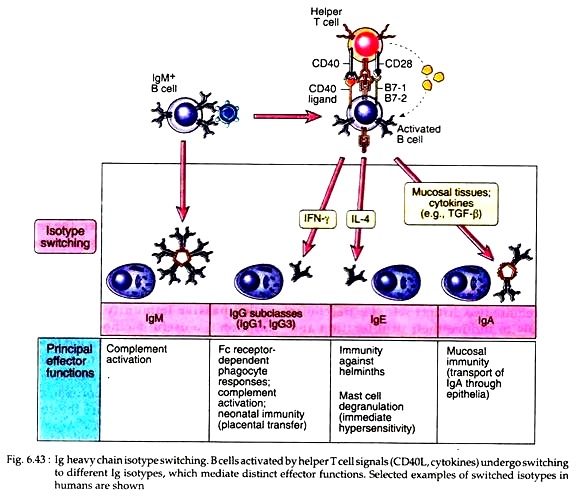
These T cells then secrete cytokines and express some ligands like CD40. This CD40 ligand then binds to CD40 on the B cells and initiates B cell proliferation and differentiation (Fig. 6.41). Cytokines (IL-2, IL-4 and IL-5) act as growth factors released by T cells. They bind to specific receptors on the B cells and increase B cell proliferation as well as differentiation.
B. Differentiation of B cell:
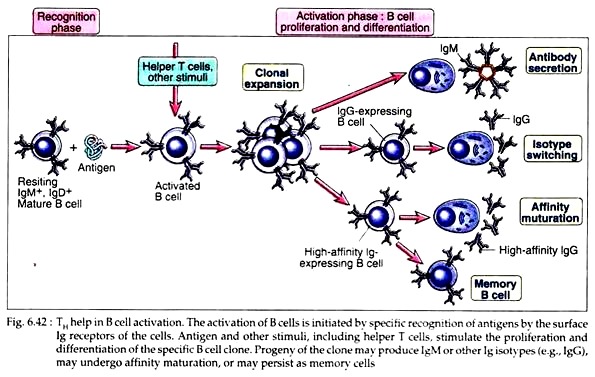
Some of the progeny of B cells that have proliferated in response to antigen and T cell help then differentiate into effector cells, plasma cells through blast cells that actively secrete antibodies.
In fact, some progeny of IgM and IgD expressing B cells then undergo the process of heavy chain iso-type switching, leading to the production of antibody with heavy chain of different classes, y, α, ɛ. Different cytokines play the most important role in regulating switching the particular heavy chain isotypes. For example, IL-4 helps the effector B cells to produce IgE antibody in all species examined.
On the other hand, some of the antigen-activated B cells do not develop into antibody secreting plasma cells, rather, they acquire the ability to survive for long periods. These are memory cells who retain the record of the antigens that they have previously encountered. These cells are capable of mounting rapid responses to subsequent introduction of antigen.
Memory B cells may remain in lymph nodes, or exit the organs and circulate in the blood and may take up residence in other lymphoid tissues. However, memory B cells are differentiable from virgin B cells by their particular cell surface marker CD4+.
The above mentioned sequences of antibody production by B cells with the help of T cells occur after the encounter with protein antigen. But non-protein antigens such as polysaccharides, lipids, glycolipids, nucleic acids etc. stimulate antibody production without requiring the help of T cells. These antigens are, therefore, termed as thymus- independent (TI).
These antigens cannot be processed and presented in association with MHC molecules and therefore they cannot be recognised by TH cells. These TI antigens are polyvalent, being composed of multiple identical antigenic epitopes. Thus, they induce maximal cross-linking of membrane Ig on specific B cells, leading to activation of the later cells.
In addition, some antigen may activate complement system by alternate pathway, generating C3d which binds to the antigen and provides second signals for B cell activation. Actually in case of B cell activation by TI antigens, there is limited or no heavy chain class switching, and memory B cell generation.
Individuals with congenital or acquired deficiencies of humoral immunity are especially susceptible to life-threatening infections with bacteria such as pneumococcus, meningococcus and haemophilus (all having cell wall with polysaccharide, i.e. TI) as there is no or little secondary immune responses.
C. Decline of Humoral Responses:
The secreted antibodies form complexes with the antigens. Such immune complexes are then cleared by macrophages with the help of complement system. Following the destruction of antigens, the specific clones of antigen-stimulated B lymphocyte undergo apoptosis. When sufficient amount of antibodies are secreted, the formed antigen-antibody complexes simultaneously bind to antigen receptors and FcƳ receptors on B cells. This binding, in turn down regulates the antibody production by plasma cells.
This phenomenon is called antibody feedback and is a physiological control mechanism in humoral immune response. Here, it should be mentioned that antibodies can also amplify antibody production by activating complement and generating C3d, a signal for B cell activation.
It is not clear when production of antibody will be stimulated or inhibited. A likely scenario is that early in humoral immune responses, IgM antibodies are involved in amplification, whereas increasing production of IgG leads to feedback inhibition.
3. Cooperation of B cells and T cells:
The cell mediated and humoral branches of the immune system assume different roles in protecting the host. The principal participants of humoral and cell-mediated immunities are the B cells and T cells, respectively.
Although humoral and cell-mediated immunities have their own distinctive features, the lymphocytes are not completely independent. In the preceding subchapters, the role of TH cells in the humoral immunity has been discussed. Here we will specifically discuss the cooperation and interaction between these two lymphocytes during adaptive immune responses.
TH Help in B cell activation:
The term “helper” T lymphocytes arose from the realisation that these cells help or stimulate B lymphocytes to produce antibodies. The helper function of T cells was discovered by experiments done in the late 1960s.
Subsequent studies established that most helper T cells are CD4+ CD8− and can recognise peptide antigens presented by class II MHC molecules. Helper T cells stimulate B cell clonal expansion, iso-type switching, affecting maturation and differentiation of B cells into memory B cells (Fig. 6.42). Different phases of T-dependent B cell activation occur in different anatomic regions within peripheral lymphoid organs.
Early events in T cell dependent B cell activation:
1. Protein antigens are recognised by specific B and T cells in peripheral lymphoid organs, and the activated cell population encounters each other at the interface of the follicles and T cell zones of lymph nodes. The T cell-B cell interaction is called a cognate interaction because it is dependent on the specific recognition of antigen and several other surface molecules on both cells.
The antigen-specific B cells first bind the naive antigen to their membrane Ig molecules, internalise and process it in endosomal vesicles. Then B cells present the peptide fragments of the antigen on their surface complexed with class II MHC molecules. Thus, the B cells themselves act as APCs in humoral immune responses to protein antigens. These peptide-MHC complexes can then be recognised by specific CD4+ helper T cells.
2. The B cells in the T cell-B cell conjugates are exposed to signals delivered by T cell surface molecules and to the highest concentration of T cell-derived cytokines. Therefore, antigen-specific B cells are the preferential recipients of T cell help and are stimulated to proliferate and differentiate.
3. Binding of antigen to membrane Ig enhances the expression of co-stimulators on the B cell membrane that increases the ability of B cell to activate helper T cell. This occurs at the same time when antigen is being processed and presented by the B cells.
The principal co-stimulators are B7-2 and B7-1 on B cell membrane. Both these molecules bind to CD28 molecule on T cell. Helper T cells can recognise peptide MHC complexes (signal 1) and co-stimulators (signal 2) and, thus, are stimulated.
4. The activated helper T cells express a surface molecule called CD40 ligand (CD40L) that can bind to its receptor CD40 on the B cell. This interaction stimulates the proliferation of B cells. Engagement of CD40 also leads to enhance expression of B7 molecules on the B cells; causing more T cell activation.
Thus, expression of B7 on B cells and CD40L on the TH cells is regulated bi-directionally with one another. This is the specificity of TH dependent B cell activation.
5. Activated T cells also secrete cytokines that act in concert with CD40L to stimulate B cell proliferation and production of antibody of different isotypes.
Cytokines serve two principal functions in antibody responses:
(i) They increase B cell proliferation and differentiation (which was initiated by CD40 signals), and
(ii) They promote switching to different heavy chain isotypes. Cytokines perform different functions in humoral immunity. Multiple cytokines including IL-2, IL-4 and IL-6 have been shown to stimulate antibody synthesis and secretion by activated B cells. Many cytokines induce heavy chain iso-type switching of the B cells (Fig. 6.43).
Late events in T cell dependent antibody responses:
Germinal centre reaction:
Within 4 to 7 days after antigen exposure, some activated B cells migrate deep into the follicles of lymph “nodes and begin to proliferate rapidly, forming a lightly stained central region of the follicles, called germinal centre.
Within these particular areas, the affinity maturation of B cell occurs. The formation of germinal centre depends on the presence of TH cells and interaction between CD40 and CD40L and is, therefore, observed only in antibody responses to helper T cell dependent protein antigens.
Affinity maturation is a process that leads to increased affinity of antibodies for a particular antigen as the T-dependent humoral responses progress. It is possible because of somatic mutation of Ig genes followed by selective survival of the B cells. The whole process requires helper T cells and CD40: CD40L interaction.
Generation of memory B cells and secondary immune response:
The generation of memory B cells and secondary immune response has already been discussed. Many of the features of secondary immune responses are due to interactions of CD4+ helper T cells and B cells. The heavy chain class switching, which is typical of secondary immune response is due to TH and their cytokines.
Actually, heavy chain class switching, affinity maturation are features usually seen in response to protein antigens because only the later can stimulate specific helper T cells.
From the above discussion it is clear that without the help of TH cells, B cell activation, proliferation, differentiation and generation of humoral immune responses will not be possible. Similarly, cell-mediated immune responses also require B cell helps.
B cell derived co-stimulators in T cell activation:
In previous subchapters, we introduced the concept that both T cell and B cell require two distinct sets of extracellular signals to induce their proliferation and differentiation into effector cells. In case of T cells, binding of peptide-MHC complexes to the TCR (T cell receptor) provides signal 1.
The second signal for T cell activation is provided by molecules on APCs, which are called co-stimulators as these molecules function together with antigen to stimulate T cells.
Among various co-stimulators, B7-1 and B7-2 are single chain glycoproteins that are expressed mainly on APCs including B lymphocytes. In B lymphocytes, engagement of the antigen receptor stimulates B7 expression, so that B cells that encounter antigen are able to activate and be helped by CD4+ helper T cells.
In this way during humoral and cell- mediated immune responses, B cell and T cell facilitate each other’s activation.